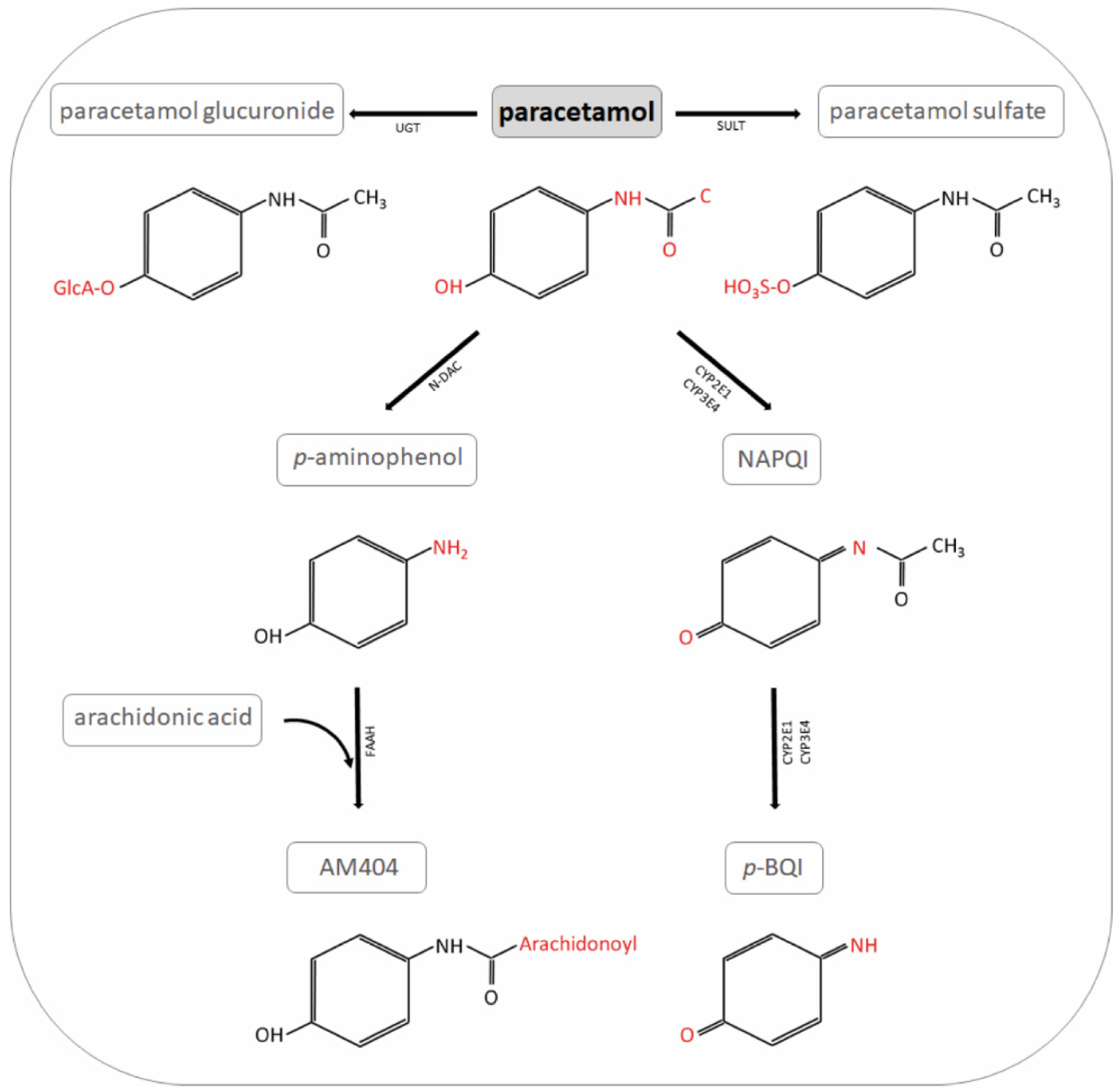
Tunable oxidative release of N-acetyl-p-benzoquinone-imine and acetamide from electrochemically derived sub-monolayer acetaminophen modified glassy carbon electrode - ScienceDirect

In-source formation of N-acetyl-p-benzoquinone imine (NAPQI), the putatively toxic acetaminophen (paracetamol) metabolite, after derivatization with pentafluorobenzyl bromide and GC–ECNICI-MS analysis - ScienceDirect

In-source formation of N-acetyl-p-benzoquinone imine (NAPQI), the putatively toxic acetaminophen (paracetamol) metabolite, after derivatization with pentafluorobenzyl bromide and GC–ECNICI-MS analysis - ScienceDirect

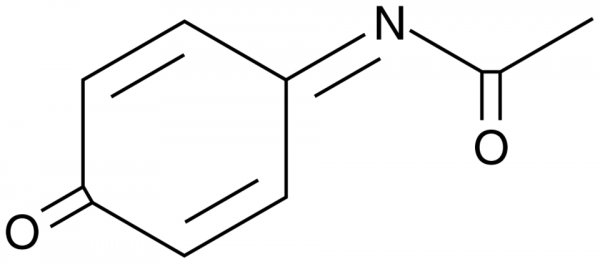
N-acetyl-p-benzoquinone imine: a cytochrome P-450-mediated oxidation product of acetaminophen. | Semantic Scholar

Sensors | Free Full-Text | Determination of Paracetamol on Electrochemically Reduced Graphene Oxide–Antimony Nanocomposite Modified Pencil Graphite Electrode Using Adsorptive Stripping Differential Pulse Voltammetry

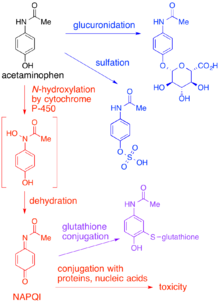
Synthesis of N-acetyl-p-benzoquinone imine (NAPQI) from acetaminophen. | Download Scientific Diagram

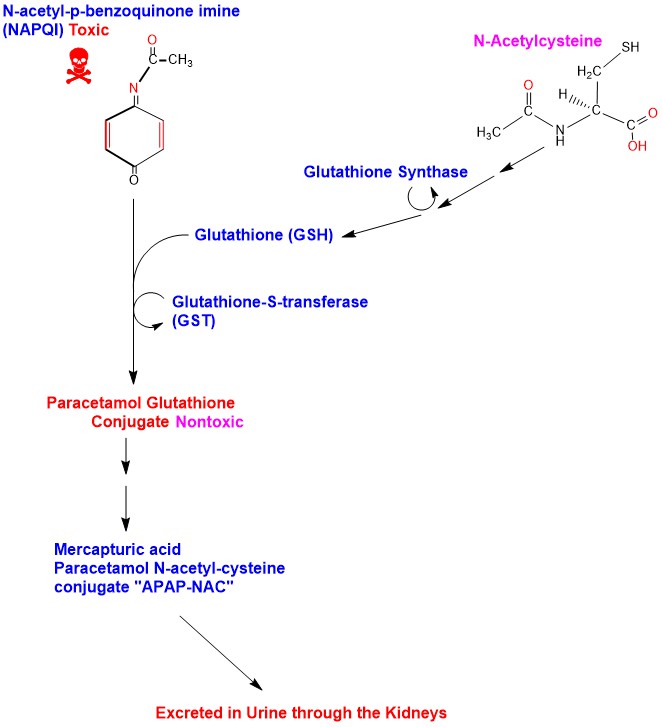
Paracetamol/acetaminophen hepatotoxicity: new markers for monitoring the elimination of the reactive N-Acetyl-p-benzoquinone imine | bioRxiv

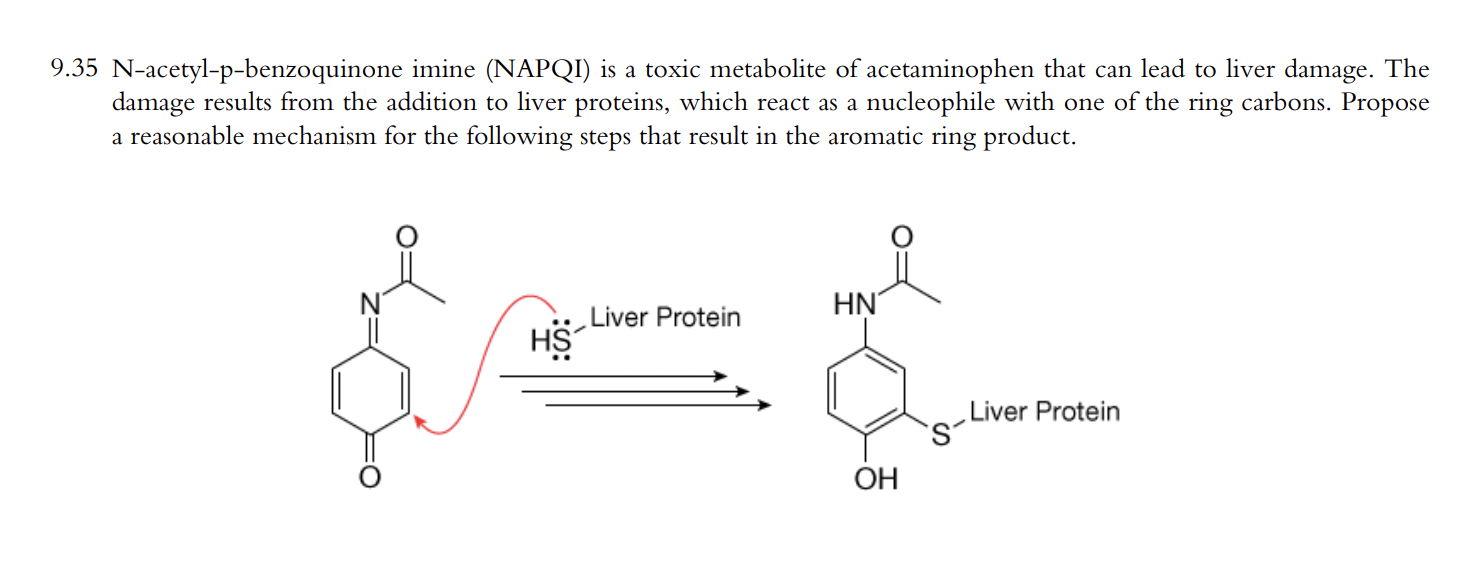
What Are the Potential Sites of Protein Arylation by N-Acetyl-p-benzoquinone Imine (NAPQI)? | Chemical Research in Toxicology
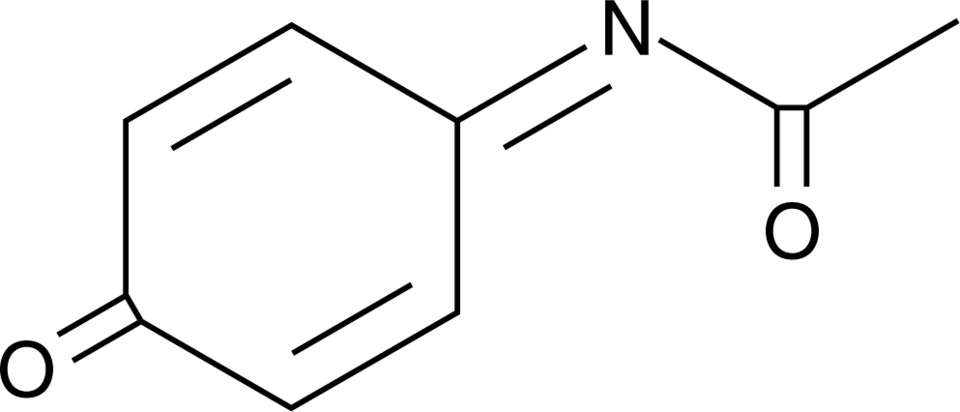
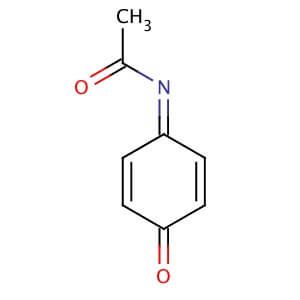
N-Acetyl-4-benzoquinone imine (NAPQI, N-Acetyl-p-benzoquinonimine, CAS Number: 50700-49-7) | Cayman Chemical