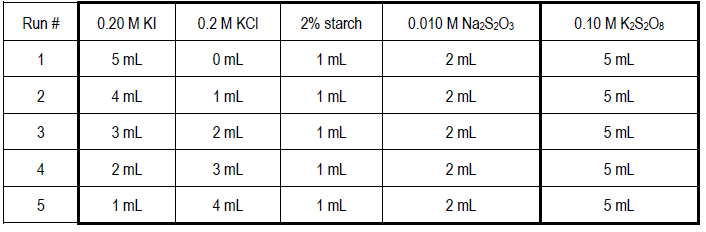
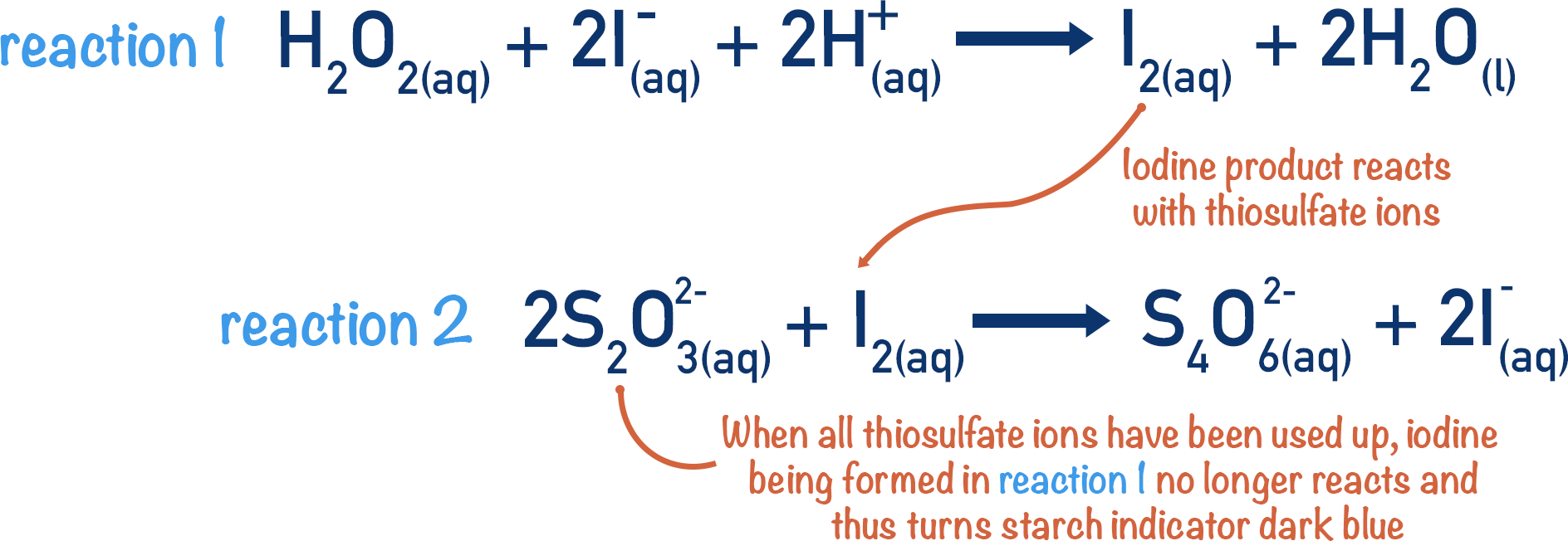
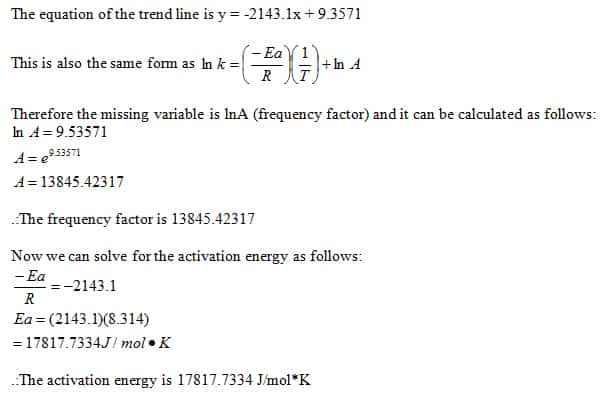PPT - In the iodine clock reaction, there are really two processes happening simultaneously. The first is a slow reaction PowerPoint Presentation - ID:39733

OneClass: In the Rate of an Iodine Clock Reaction experiment, the following data were collected by a ...

SOLVED: Reaction Rates Concentrations: Effect of Reactant Concentrations Demo: "Iodine Clock" Reaction Iodate Variation IO3- + 3 HSO3- â†' 1-+ 3 SO42- + 3 H2O (slow) IO3- + 5I- + 6 H+




![Solved The iodine clock reaction Test [KIO3] (M) [NaHSO3] | Chegg.com Solved The iodine clock reaction Test [KIO3] (M) [NaHSO3] | Chegg.com](https://media.cheggcdn.com/media/4ea/4ea146b7-b7f4-4915-88de-6a4373a115e2/phpLZvxWR)