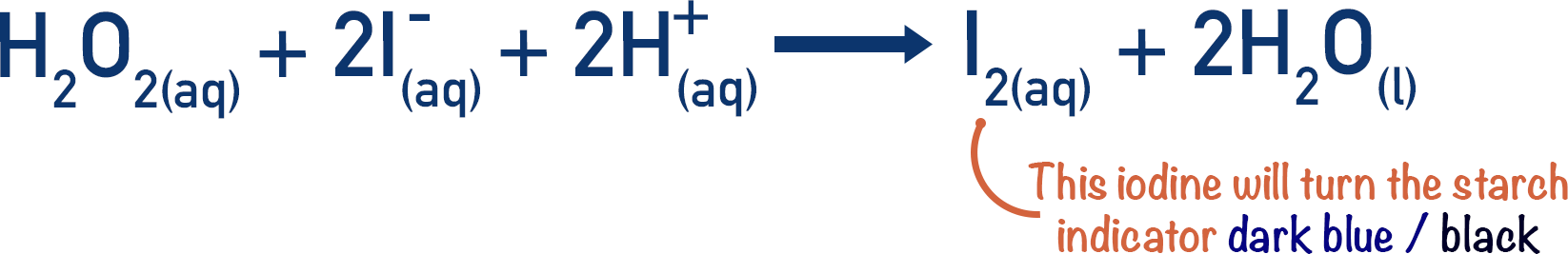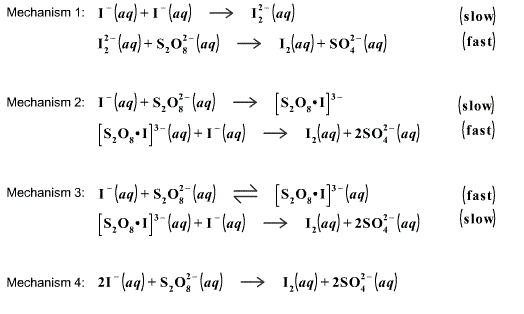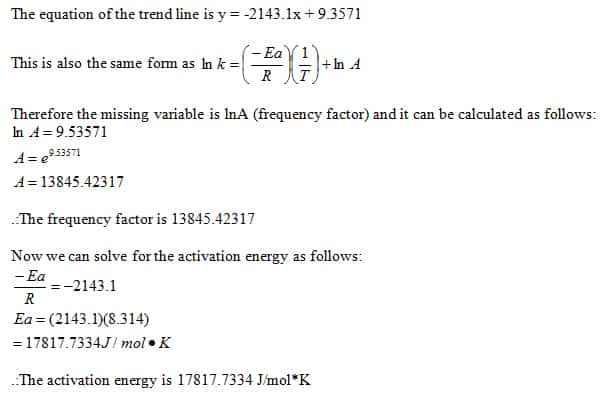Iodine+Clock+Part+A+Sp21 - Iodine Clock Reaction Part A Stephanie R. Geggier New York University, - Studocu

OneClass: In the Rate of an Iodine Clock Reaction experiment, the following data were collected by a ...

SOLVED: Reaction Rates Concentrations: Effect of Reactant Concentrations Demo: "Iodine Clock" Reaction Iodate Variation IO3- + 3 HSO3- â†' 1-+ 3 SO42- + 3 H2O (slow) IO3- + 5I- + 6 H+

PPT - In the iodine clock reaction, there are really two processes happening simultaneously. The first is a slow reaction PowerPoint Presentation - ID:39733

Exp16calcs W - lab report - Chemical Kinetics: The Iodine-Clock Reaction: S 2 O 82 −(aq) + 2 I−(aq) - Studocu